For Researchers and Companies iPS Cell Stock Project
Overview of iPS Cell Stock Project
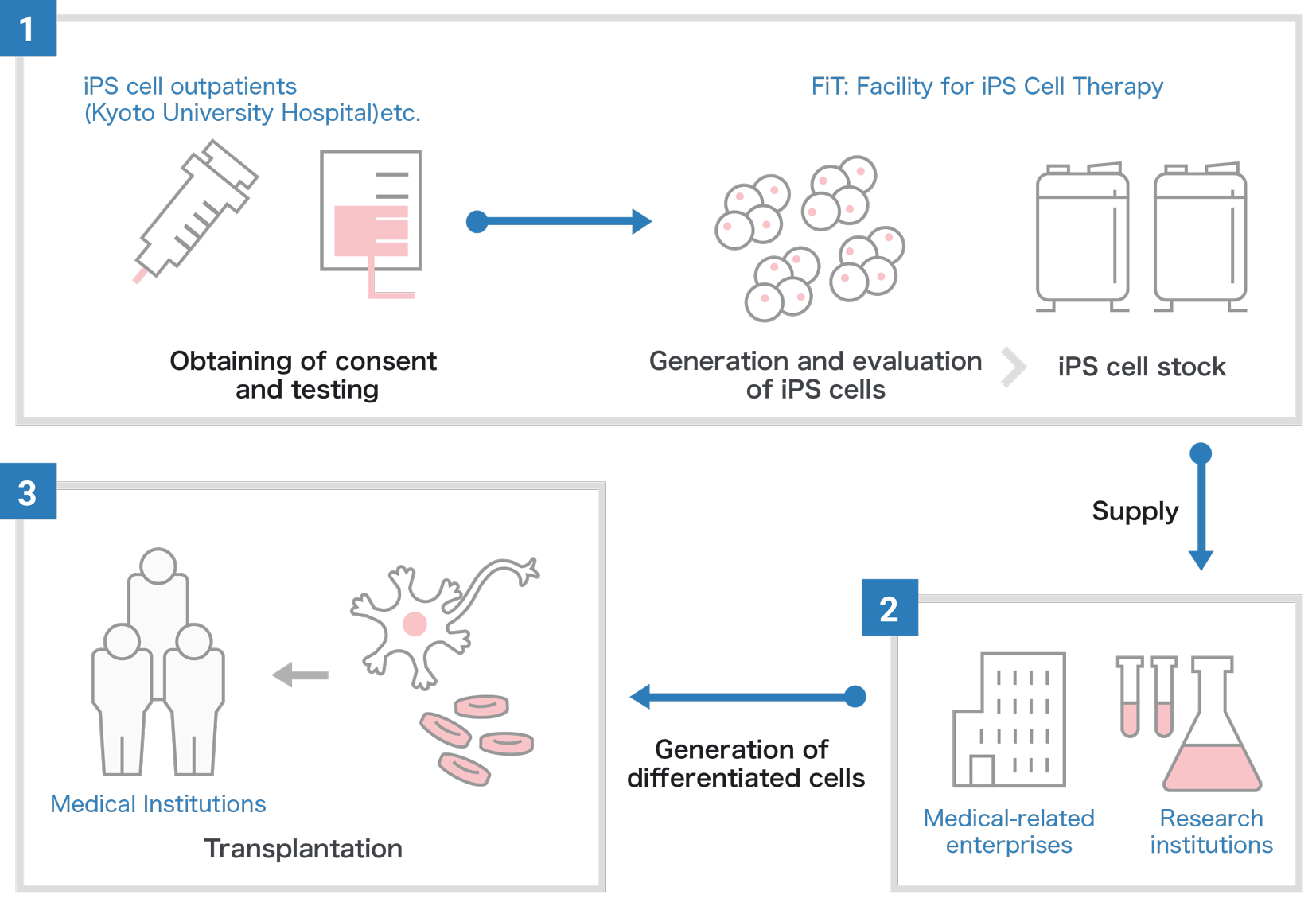
iPS Cell Stock Project
The iPS Cell Stock Project is collecting blood from HLA (human leukocyte antigen)- homozygous donors, because these cells are expected to lower the risk of immune rejection in transplants after donor-patient matching. These blood cells are reprogrammed into iPS cells for regenerative medicine at FiT (Facility for iPS Cell Therapy). The stocked iPS cells undergo strict quality control for clinical use and are distributed upon request to hospitals or research centers both in Japan and overseas for the purpose of treating patients suffering from intractable diseases. It is estimated that the current iPS cell stock at FiT could match 40 percent of the Japanese population.
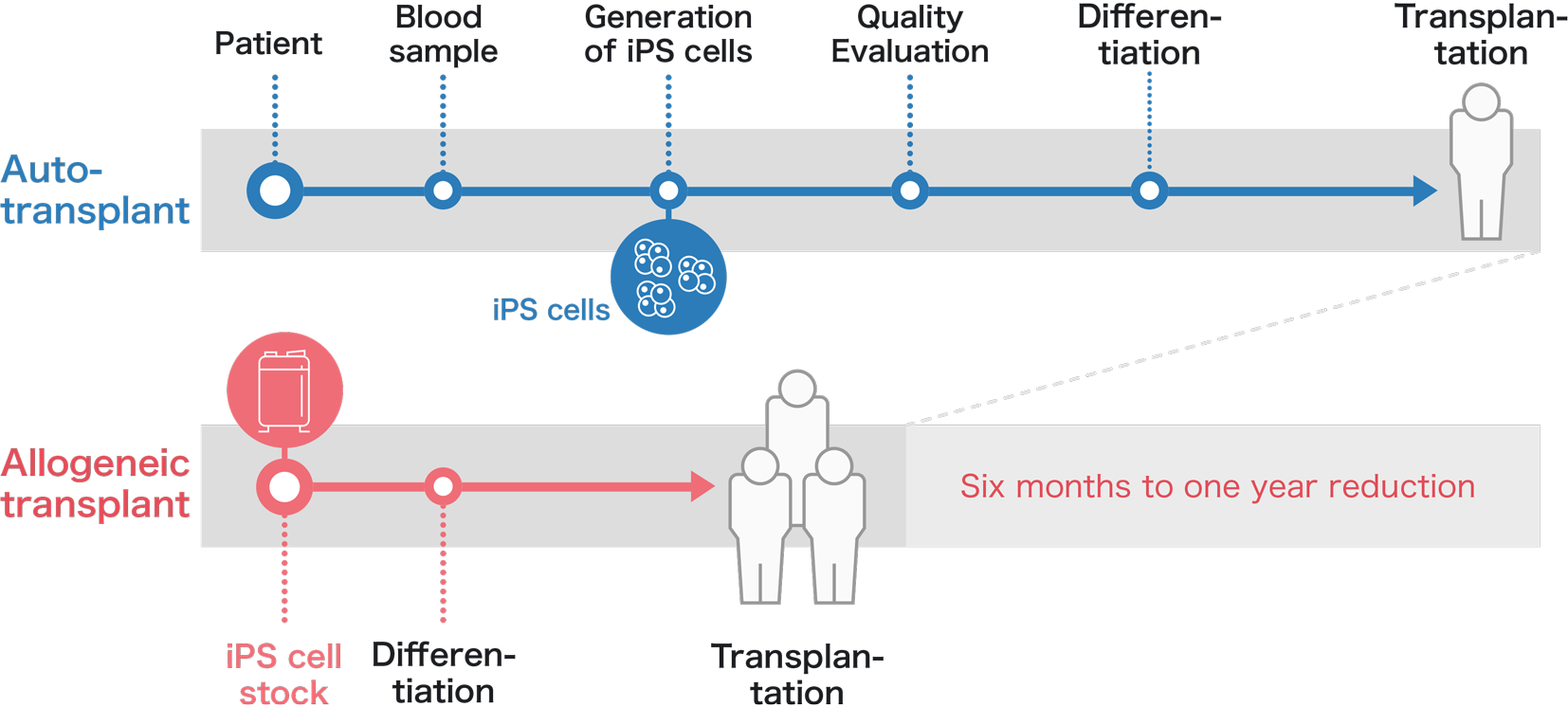
Ideally autologous iPS cells will be used for the transplantation. To do so requires the patient's own cells such as blood be reprogrammed to iPS cells and differentiated into the cell type used for the transplantation. Each step involving cell manipulation, from reprogramming to differentiation, requires strict quality control.
However, the iPS Cell Stock Project uses allogeneic iPS cells, which allows the cells to have passed quality control before the cells are demanded, saving time and cost compared to autologous transplants.
The CiRA Foundation has inherited the cell-manufacturing technologies and quality-control systems at CiRA and also inherited FiT, which will continue to produce and stock iPS cells.
This project has been supported by the organizations listed below, who have helped in the donor recruitment and blood collection.
Related Information
- For Researchers and Companies
- Research Materials(Bioresources)
- iPS Cell Stock Project
- How to Use the Stock
